Influence of Interval Period on Root Resorption Due To Orthodontic Force
Junya Nagata, Kunihiko Nojima, Yasushi Nishii, Kenji Sueishi, Takaki Yamazaki and Yasuo Miake
1Department of Orthodontics, Tokyo Dental College, 1-2-2 Masago, Mihama-ku, Chiba 261-8502, Japan
2Department of Histology and Developmental Biology Tokyo Dental College 2-9-18 Misakityou, Tiyoda-ku, Tokyo 101-0061, Japan
- *Corresponding Author:
- Junya Nagata
Department of Orthodontics, Tokyo Dental College
1-2-2 Masago, Mihama-ku, Chiba261-8502, Japan
Tel: 81-043-270-3966
E-mail: nagatajunya@tdc.ac.jp
Received date: February 09, 2016; Accepted date: February 29, 2016; Published date: March 03, 2016
Citation: Nagata J. Influence of Interval Period on Root Resorption Due to Orthodontic Force. J Orthod Endod. 2016, 2:1.
Abstract
We used 3D laser microscopy to conduct a quantitative analysis and to compare the changes in root resorption due to orthodontic force with or without an interval period after development of root resorption. Experimental device with closed coil spring extending from the maxillary incisor to the first molar was used. 20 rats were divided into 4 groups; 20 rats underwent 50-gf traction for 1 week and were subsequently divided into 4 groups. 50-gf traction group was sacrificed immediately after undergoing 50-gf traction. Interval group underwent 50-gf traction and was sacrificed after an interval of 2 weeks. Continuous traction group underwent 50- gf traction and was sacrificed after undergoing further 10-gf traction for 2 weeks. Post-interval traction group underwent 50-gf traction followed by an interval of 2 weeks and were sacrificed after undergoing further 10-gf traction for 2 weeks. The surface area and volume of root resorption craters and the percentage of tooth root surface area occupied by these craters were measured using 3D laser microscopy. Traction of 10 gf caused no significant differences in the degree of tooth movement between the groups. Quantitative analysis showed no significant differences between the 50 gf traction and the interval groups; however, there was a significant difference between the continuous traction and post-interval traction groups. Results indicated that when root resorption occurs, traction should be temporarily interrupted and resumed after an interval period.
Keywords
Orthodontic force; Root resorption; Interval period; 3D laser microscopy
Introduction
There are many accidents to happen during orthodontic treatment. The root resorption is a big problem in orthodontist. Many risk factors are related to onset of root resorption [1-10]. However, root resorption may develop even if it was treated avoiding a risk. When root resorption occurs, the resulting resorption craters should be allowed to repair before performing any further procedures. Cheng et al. [11] monitored the repair of root resorption craters in human molars over 4-8 weeks and found that longer repair periods resulted in better root surface repair. Gonzales et al. [12] monitored the repair of root resorption craters in rat molars over 2–16 weeks and found 67% repair of tooth resorption after 16 weeks. Levander et al. [13] observed teeth in which severe root resorption occurred in patients who continued orthodontic treatment and in whom orthodontic forces were not applied to these teeth for a further 2–3 months; they found that root resorption was suppressed in patients who did not undergo orthodontic tooth movement. In this way, in clinical practice, it is often necessary to move a tooth with root resorption. In such cases, it is suggested that an optimal or light force should be applied to these teeth, avoiding the application of heavy force that causes root resorption, and these teeth should be regularly monitored radiographically. However, few basic studies have examined changes in root resorption craters due to differences in methods of orthodontic force application after root resorption. The present study aimed to use three-dimensional (3D) laser microscopy to perform a quantitative analysis and to compare changes in root resorption craters due to orthodontic force with or without an interval period after development of root resorption.
Materials and Method
Experiments were performed in 20 7-week-old male Wistar rats. This study was approved by the Animal Experiment Committee of Tokyo Dental College (approval number: 242801). Animals had free access to powdered chow (Oriental Yeast Co., Ltd., Tokyo, Japan) and tap water throughout the duration of the experiment.
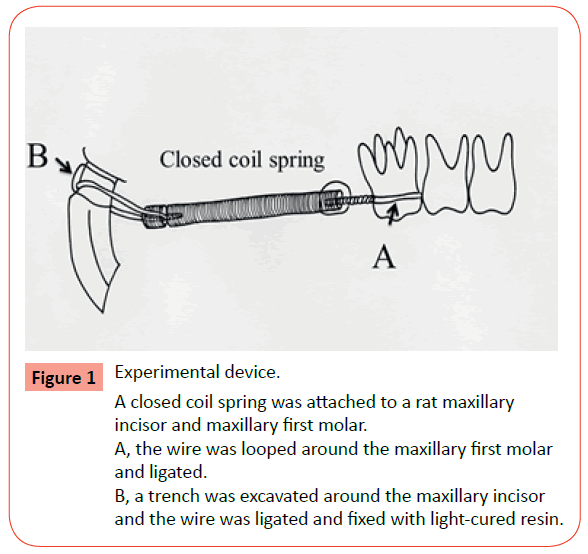
Orthodontic devices were placed after rats were anesthetized with an intraperitoneal injection of pentobarbital sodium at a dose of 64.8 g/ kg body weight (Kyoritsu Seiyaku Co., Ltd, Tokyo, Japan). The experimental device used was a 0.08 × 0.30-inch closed coil spring (3M Unitek, Oklahoma, USA). Ligature wires were attached to both ends of the spring, and the spring was ligated to the maxillary first molars. A dental turbine hand piece was used to excavate a trench around the entire circumference of the maxillary incisors, following which the ligature wires attached to the spring were ligated to the incisors (Figure 1). Light-cured resin (GC Co. Ltd., Tokyo, Japan) was used every 7 days to prevent detachment of the device from the incisors [7].
Figure 1 Experimental device.
A closed coil spring was attached to a rat maxillary
incisor and maxillary first molar.
A, the wire was looped around the maxillary first molar
and ligated.
B, a trench was excavated around the maxillary incisor
and the wire was ligated and fixed with light-cured resin.
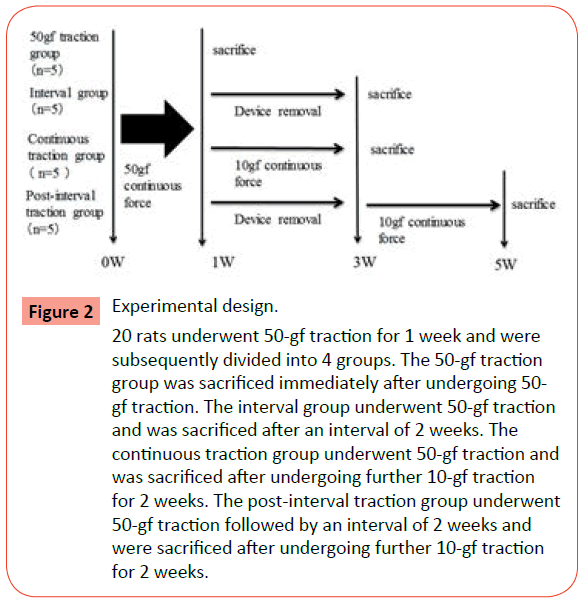
The 20 rats were randomly allocated to 4 treatment groups (n = 4 / group): 20 animals underwent 50-gf traction for 1 week, after which they were divided into 4 groups according to their subsequent treatment. In the 50-gf traction group, experimental specimens were harvested immediately after the traction period. In the interval group, the device was removed and experimental specimens were harvested 2 weeks later. In the continuous traction group, 10-gf traction was applied for a further 2 weeks before experimental specimens were harvested. In the postinterval traction group, the device was removed, and after a 2-week interval, 10-gf traction was applied for a further 2 weeks before experimental specimens were harvested (Figure 2). The degree of tooth movement was measured before and after of 10- gf and 50-gf traction period. The distance between the cervical points of the lingual surface of the maxillary incisor to the mesial surface of the maxillary first molar was measured using digital Vernier calipers (Mitutoyo Corporation, Kanagawa, Japan). The degree of tooth movement was measured 5 times and the mean values were calculated.
Figure 2 Experimental design. 20 rats underwent 50-gf traction for 1 week and were subsequently divided into 4 groups. The 50-gf traction group was sacrificed immediately after undergoing 50- gf traction. The interval group underwent 50-gf traction and was sacrificed after an interval of 2 weeks. The continuous traction group underwent 50-gf traction and was sacrificed after undergoing further 10-gf traction for 2 weeks. The post-interval traction group underwent 50-gf traction followed by an interval of 2 weeks and were sacrificed after undergoing further 10-gf traction for 2 weeks.
Rats were sacrificed after anesthesia with diethyl ether (Shouwakagaku, Tokyo, Japan), and the maxillary first molars were harvested en bloc along with the surrounding tissue. Harvested specimens were immersed in 10% formalin for 1 day. Soft tissues other than the periodontal ligament (PDL) were removed, following which the PDL was removed after treating with 10% sodium hypochlorite for 20 minutes. The distolingual root was severed and used as the experimental specimen.
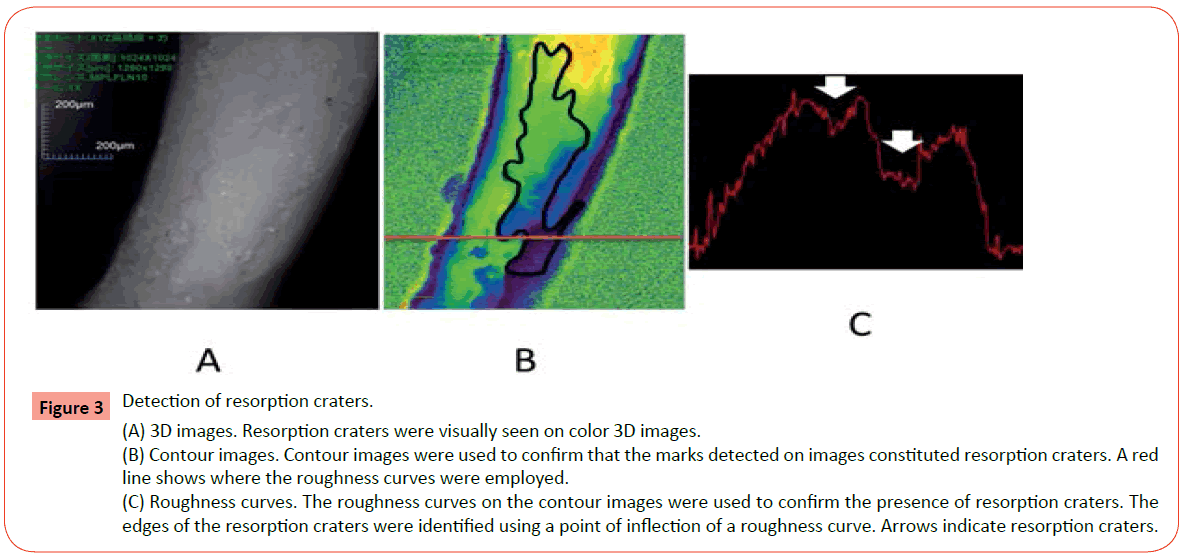
Measurements were performed using a 3D laser measuring microscope (Olympus, Tokyo, Japan), and profiles of the pressure sides of the distolingual roots of the maxillary first molar were generated. Color imaging was used to identify the root resorption craters; the differences in crater shape were assessed visually and the root resorption craters were delineated (Figure 3A). Contour images were used to confirm the constituted resorption craters. In addition, roughness curves were observed at the red line portion in the contour image (Figure 3B), and resorption craters were identified using the points of inflection of the roughness curves (Figure 3C). The following measurements were performed on the pressure side of the root surface.
Figure 3 Detection of resorption craters. (A) 3D images. Resorption craters were visually seen on color 3D images. (B) Contour images. Contour images were used to confirm that the marks detected on images constituted resorption craters. A red line shows where the roughness curves were employed. (C) Roughness curves. The roughness curves on the contour images were used to confirm the presence of resorption craters. The edges of the resorption craters were identified using a point of inflection of a roughness curve. Arrows indicate resorption craters.
The surface areas of root resorption craters (RRA) were measured using existing software.
The depth of resorption craters between the mid-points of the highest and the lowest areas of the craters was measured, and the volume of the root resorption craters (RRV) was calculated using existing software. When discrete resorption craters were present, the volume of each crater was calculated and summed.
The percentage of the tooth root surface area occupied by root resorption craters (RRA %) was calculated using the following equation:
RRA (%) = RRA (μm2) / root surface area of the pressure side (μm2) × 100
Unpaired t-test was used as a comparison test, to see in RRA, RRV and RRA (%) between 50-gf traction group and interval group, and between continuous traction group and post-interval traction group. Also the Unpaired t-test was used to see the mean degree of tooth movement at the time of 10-gf traction and 50-gf traction. Differences with ≤5% level of significance were considered statistically significant.
Results
Degree of tooth movement
The mean distance that the teeth moved under 50-gf traction was 0.2 ± 0.06 mm all 4 groups after the 1 week of 50-gf traction. There was no significant difference between 4 groups. The mean degree of tooth movement at the time of 10-gf traction of continuous traction group and post-interval traction group was 0.39 ± 0.05 mm and 0.40 ± 0.06 mm. There was no significant difference between continuous traction group and post-interval traction group in the mean degree of tooth movement caused by 10-gf traction for 2 weeks.
RRA and RRV
RRA: The measurements in the 50-gf traction and interval groups were not significantly different. There was significant difference between continuous traction group and post-interval traction group. The continuous traction group showed the highest degree of root resorption.
RRV: The measurements in the 50-gf traction and interval groups were not significantly different. There was significant difference between continuous traction group and post-interval traction group. The continuous traction group showed the highest degree of root resorption.
RRA (%): The continuous traction group showed the highest degree of root resorption. There were significant differences between 50-gf traction group and interval group, continuous traction group and post-interval traction group (Table 1).
| Groups | RRA (x 10 4111.M2) | RRV (x 106pm3) | RRA(%) | |||
|---|---|---|---|---|---|---|
| 50gf traction group | 10.2±2.3 | N.S. | 5.3±0.9 | N.S. | 14.6±1.7 | * |
| Interval group | 9.2±0.8 | 4.2±0.8 | 11.6±0.4 | |||
| Continuous traction group | 20.3±2.6 | * * | 13.5±1.6 | ** | 28.2±1.9 | ** |
| Post -interval traction group | 14.5±1.1 | 9.1±0.5 1 | 15.1±1.3 |
The continuous traction group showed the highest degree of root resorption. There was significant difference between continuous traction group and post-interval traction group.
Table 1: Experimental results.
Discussion
Experimental methods
We performed a comparative investigation of changes in root resorption craters due to orthodontic force with or without an interval period after the development of root resorption. Therefore, it is important to determine the light orthodontic force to apply to rat molars and the force causing root resorption. Kondo [9] reported that the optimal force was 80 g/cm2 for maxillary canines in cats, which was calculated from capillary blood pressure. Kirino et al. [7] used 10 gf as the optimal force on the PDL of rat maxillary molars, which reportedly corresponds to a force of 80 g/cm2. Kohno et al. [10] also reported that the optimal orthodontic force for rat maxillary molars is < 10 gf. Studies investigating the effect of different orthodontic forces on root resorption in rat molars have also reported that little root resorption occurred when 10 gf was applied and that a higher degree of resoption occurred at 50 gf and 100 gf [8]. Gonzales et al. [12] used a 50-gf force to cause root resorption, which reportedly corresponds to a 1000-gf force in human molars [14]. Based on data from these studies, we used 10 gf as a light orthodontic force and 50 gf as a force resulting in root resorption in the present study.
Rat maxillary molars have 5 roots, and several studies that measured root resorption used the pressure side on the mesial root [15-17]. Gonzales et al. [8] moved rat maxillary molars and investigated the differences in the degree of resorption in each root; they found that in terms of resorption craters, more resorption occurred on the pressure side of distal roots than on the pressure side of central roots. Other studies using the pressure side of distal roots to measure root resorption craters have also been reported [11-18]. Therefore, we used the pressure side of the distolingual roots for evaluation on the basis of the fact that a greater effect of root resorption enables easier quantitative measurement and observation of root resorption craters.
To evaluate root resorption crater formation, numerous studies have used various methods such as tissue slices, transmission electron microscopy, scanning electron microscopy (SEM), and 3D laser microscopy [8-19]. Chan et al. [19] reported that because technical skills are necessary to produce tissue slices and the cut portion is lost in the process of obtaining tissue slices, tissue slices is unreliable for measuring root resorption craters. Fujimura et al. [15] used SEM to measure the percentage of tooth root surface area occupied by root resorption craters. SEM is useful for quantitative measurement of root resorption because of its high spatial resolution. Gonzales et al. [8] used 3D laser measuring microscopy to measure the percentage of tooth root surface area occupied by root resorption craters and crater depth, volume, and roughness. The 3D laser microscope is used to observe from one direction. If better machines are used to observe from many directions a more accurate judgment of the root resorption fossa, thus, a more correct result is expected. However the present study used 3D laser microscopy to evaluate root resorption because this method does not necessitate surface treatment of the sample, has many measurement items, provides spatial resolution, and is extremely useful for evaluating tertiary structure.
Root resorption
In the present study no significant differences in RRA or RRV were observed between the 50-gf traction and interval groups. Repair of root resorption craters is reportedly time-dependent [12]. The amount of repair cementum increases over the first 4 weeks, finally reaching a stable phase [20]. Gonzales et al. [9] reported that repair becomes more active after 4 weeks, with 67% of repair achieved after 16 weeks. Therefore, this study suggests that repair of root resorption tends to occur in 2 weeks, but a longer period is necessary to make a significant difference.
Although in the present study no significant differences in RRA or RRV were observed between the 50-gf traction and interval groups, RRA (%) was significantly different between these groups.This difference may have resulted from the fact that rats in the 50-gf traction group were 8 weeks old, whereas those in the interval group were 10 weeks old; an increase in the total root surface area was seen because of growth in the interval group.
The present study, the post-interval traction groups and the continuous traction group exhibited significant differences in RRA, RRV and RRA (%). There was no significant difference between 50-gf traction group and interval group, however there was significant difference between continuous traction group and post-interval traction group. In addition, Brudvik et al. [21] showed that the cementum adjacent to hyalinized areas of the PDL is “marked” by this contact and that osteoclasts attack this marked cementum when the PDL area is repaired. In the Postinterval traction group, repair of root resorption, although insignificant, was seen. It can be assumed that the resorption fossa was repaired, thus resulting in a significant difference. This finding suggests that it may be necessary to interrupt tooth movement temporarily to allow repair of root resorption craters and that tooth movement after root resorption increases the risk of further root resorption.
Tooth movement
Tooth movement can be divided into three phases: initial displacement, lag, and proportional. In growing rats (aged 6 weeks), application of a 10-gf force results in rapid tooth movement up to day 3, followed by a lag phase ending before day 7, which in turn is followed by further movement. It has been reported that when a 40-gf force is applied, the lag phase lasts until day 7 [22]. In the present study, no significant difference in the degree of tooth movement was observed between when 50- gf traction and no significant difference in the degree of tooth movement were observed between continuous traction and postinterval traction groups. We consider that the tooth movement in the continuous traction group and post-interval traction groups were not different because of the active resorptive function of osteoclasts, although the continuous traction group may have been affected by hyaline degeneration. However, considerable root resorption occurred in the continuous traction group. Therefore, it may be necessary to move teeth after allowing repair of root resorption to avoid further risk of tooth injury due to orthodontic treatment.
Conclusion
Our results verified using 3D laser measuring microscopy demonstrated that when root resorption occurs, traction should be temporarily interrupted and resumed after an interval period.
Conflict of Interest
The authors declare that no competing interests exist.
References
- Reitan K (1974) Initial tissue behavior during apical root resorption. Angle Orthod 44:68-82.
- Maltha JC, Van Leeuwen EJ, Dijkman GE, Kuijpers-JagtmanAM (2004) Incidence and severity of root resorption in orthodontically moved premolars in dogs. Ortho Craniofac Res 4:115-121.
- Mirabella AD, Artun J (1995) Risk factor for apical root resorption of maxillary anterior teeth in adult orthodontic patients. Am J OrthodDentofacialOrthop108:48-55.
- Bridges T, King G, Mohammed A (1988)The effect of age on tooth movement and mineral density in the alveolar tissues of the rat. Am J OrthodDentofacialOrthop 93:245-250.
- Mavragani M, Bøe OE, Wisth PJ, Selvig KA (2002) Changes in root length during orthodontic treatment: advantages for immature teeth. Eur J Orthod 24:91-97.
- Chan E, Darendeliler MA (2005) Physical properties of root cementum: Part 5. Volumetric analysis of root resorption craters after application of light and heavy orthodontic forces. Am J OrthodDentofacialOrthop 127:186-195.
- Kirino Y, Tsuchiya T, Kurihara S, Chiba M, Miura F (1991) A study of tooth movement with super-elastic force. J JpnOrthodSoc 50:315-324.
- Gonzales C, Hotokezaka H, Yoshimatsu M, Joseph H, Yozgatian M, et al. (2008) Force magnitude and duration effects on amount of tooth movement and root resorption in the rat molar. Angle Orthod 78:502-509.
- Kondo K (1969) A study of blood circulation in the periodontal membrane by electric impedance plethysmography. KokubyoGakkaiZasshi 36:20-42.
- Kohno T, Matsumoto Y, Kanno Z, Warita H, Soma K (2002) Experimental tooth movement under light orthodontic forces: rates of tooth movement and changes of the periodontium. J Orthod 29:129-135.
- Cheng LL, Türk T, Elekdag-Türk S, Jones AS, Yu Y, et al. (2010) Repair of root resorption 4 and 8 weeks after application of continuous light and heavy forces on premolars for 4 weeks: A histology study. Am J OrthodDentofacialOrthop 136:727-734.
- Gonzales C, Hotokezaka H, Darendeliler MA, Yoshida N (2010) Repair of root resorption 2 to 16 weeks after the application of continuous forces on maxillary first molars in rats: A 2- and 3-dimensional quantitative evaluation. Am J OrthodDentofacialOrthop 137:477-485.
- Levander E, Malmgren O, Eliasson S (1994) Evaluation of root resorption in relation to two orthodontic treatment regimes. A clinical experimental study. Eur J Orthod 16:223-228.
- Ren Y, Maltha JC, Kuijpers-Jagtman AM (2004)The rat as a model for orthodontic tooth movement a critical review and a proposed solution. Eur J Orthod 26:483-490.
- Fujimura Y, Kitaura H, Yoshimatsu M, Eguchi T, Kohara H, et al. (2009) Influence of bisphosphonates on orthodontic tooth movement in mice. Eur J Orthod 31:572-577.
- Kumasako-Haga T, Konoo T, Yamaguchi K, Hayashi H (2009) Effect of 8-hour intermittent orthodontic force on osteoclasts and root resorption. Am J OrthodDentofacialOrthop 135: e1-8.
- Baysal A, Uysal T, Ozdamar S, Kurt B, Kurt G, et al. (2010) Comparisons of the effects of systemic administration of L-thyroxine and doxycycline on orthodontically induced root resorption in rats. Eur J Orthod 32:496-504.
- Gonzales C, Hotokezaka H, Karadeniz EI, Miyazaki T, Kobayashi E, et al. (2011) Effects of fluoride intake on orthodontic tooth movement and orthodontically induced root resorption.Am J OrthodDentofacialOrthop139:196-205.
- Chan E, Darendeliler MA (2004) Exploring the third dimension in root resorption. OrthodCraniofac Res 7:64-70.
- Owman-Moll P, Kurol J, Lundgren D (1995) Repair of orthodontically induced root resorption in adolescents. Angle Orthod 65:403-408.
- Brudvik P, Rygh P (1995) Transition and determinants of orthodontic root resorption-repair sequence. Eur J orthod 17:177-188
- Kyomen S, Tanne K (1997) Influences of aging changes in proliferative rate of PDL cells during experimental tooth movement in rats. Angle Orthod 67:67-72.
Open Access Journals
- Aquaculture & Veterinary Science
- Chemistry & Chemical Sciences
- Clinical Sciences
- Engineering
- General Science
- Genetics & Molecular Biology
- Health Care & Nursing
- Immunology & Microbiology
- Materials Science
- Mathematics & Physics
- Medical Sciences
- Neurology & Psychiatry
- Oncology & Cancer Science
- Pharmaceutical Sciences